Organoelement
Ito Research Lab
We aim to develop globally competitive
researchers as pioneers of the academic field,
achieving the top-class research method and data.
Hajime Ito's Laboratory
The major research interests of this laboratory are the development of new synthetic reactions, the exploration of new organic materials, and the discovery of unprecedented phenomena in chemistry. Our current research projects include (1) the copper-catalyzed reactions of diboron for selective organoboron synthesis; (2) iridium-catalyzed C-H borylation reactions; (3) the mechanochromic luminescence of organogold complexes; and (4) the development of new enantio-convergent reactions for the efficient synthesis of optically active compounds. The environment of this research group is designed to inspire all of the group members, and to cultivate and nurture their creativity by providing an interesting mixture of different elements of chemical research, with research topics ranging from organic synthesis to the development of nanomaterials. In this laboratory, our students and young researchers will learn many practical techniques that they will be able to apply in various fields of chemical research, including organic synthesis, coordination chemistry, the development of homogeneous catalysts, asymmetric synthesis, luminescent materials, and the development of organic materials based on nanoarchitectures using a bottom-up approach.

PUBLICATIONS
-
Mechanochemistry-directed ligand design for enhanced reactivity and enantioselectivity in solvent-less palladium-catalyzed conjugate arylations
Kondo, K.; Shoji, H.; Kubota, K.*; Ito, H.* RSC Mechanochem. 2026, Just Accepted
DOI :10.1039/D6MR00032K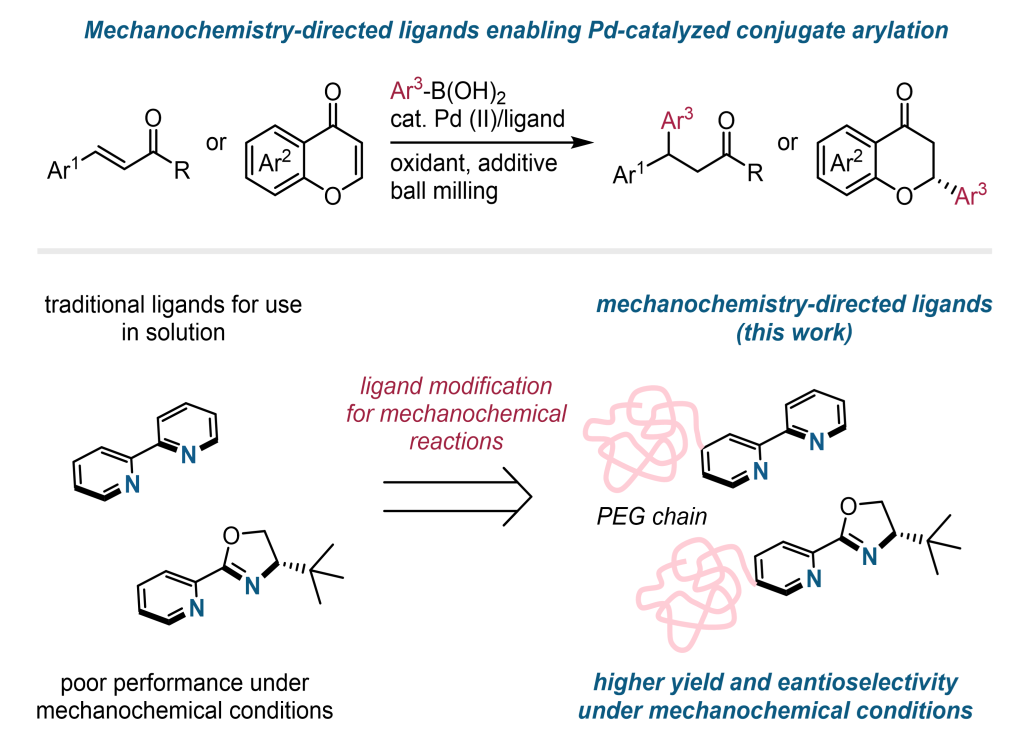
-
Amano-PS-IM-Catalyzed Mechanochemical Enzymatic Acylative Kinetic Resolution of Secondary Alcohols
Kubota, K.*; Kamakura, Y.; Takeda, T.; Ito, H.* RSC Mechanochem. 2026, Just Accepted
DOI :10.1039/D6MR00028B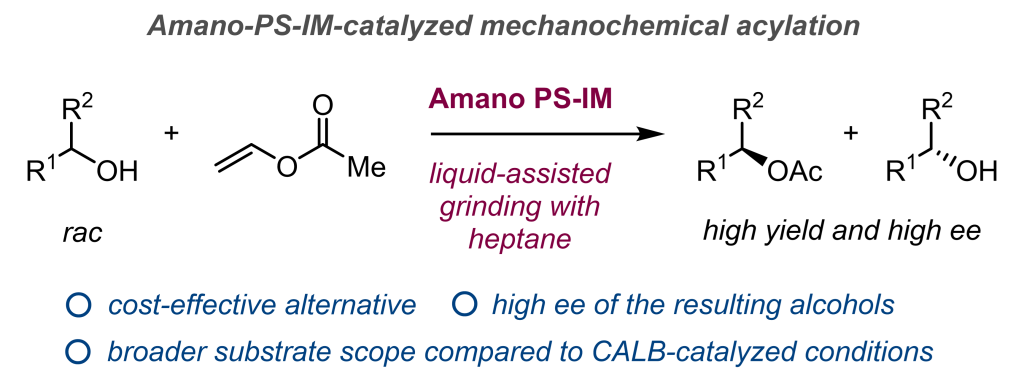
-
Breaking the Immiscibility Barrier: A Mechanochemical Approach to Enable the Direct Glycosylation of Native Sugars with Hydrophobic Aliphatic Alcohols
Kubota, K.*; Kamakura, Y.; Takeda, T.; Jiang, J.; Manabe, Y.; Fukase, K.; Ito, H.* J. Am. Chem. Soc. 2026, Just Accepted
DOI :10.1021/jacs.6c04094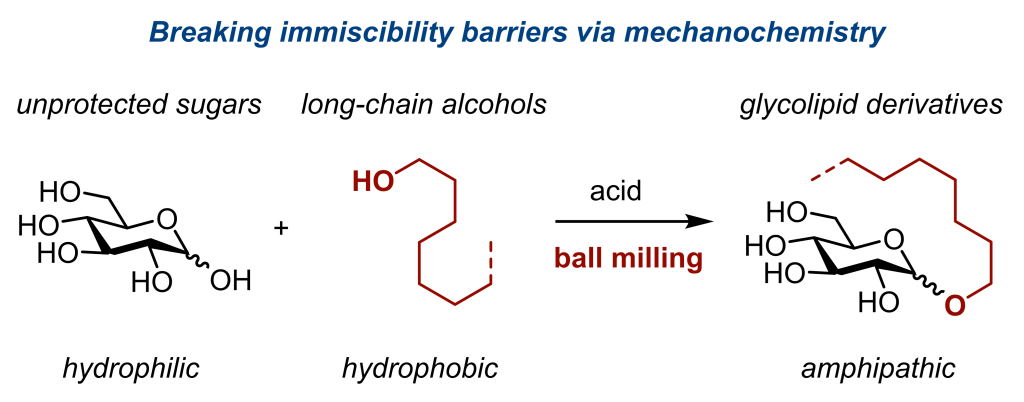
Contactus
- Professor Dr. Hajime Ito
-
E-mail: hajitoeng.hokudai.ac.jp
